Results All three pretreatments resulted in significant solubilization of lignin and xylan, with the CELF pretreatment solubilizing the majority of both biopolymer categories. Lignins recovered from the pretreatments were characterized for polysaccharide content, more » molar mass distributions, β-aryl ether content, and response to depolymerization by thioacidolysis. For each of the feedstocks, the pretreatments were assessed for their impact on lignin and xylan solubilization and enzymatic hydrolysis yields as a function of enzyme loading. These include co-solvent-enhanced lignocellulosic fractionation (CELF), pretreatment with an ionic liquid using potentially biomass-derived components (cholinium lysinate or ), and two-stage Cu-catalyzed alkaline hydrogen peroxide pretreatment (Cu-AHP). « lessĪbstract Background In this work, three pretreatments under investigation at the DOE Bioenergy Research Centers (BRCs) were subjected to a side-by-side comparison to assess their performance on model bioenergy hardwoods (a eucalyptus and a hybrid poplar). In conclusion, this study provided a new approach to effectively isolate depolymerized lignin from lignocellulose in a less condensed form for boosting its downstream valorization. Preliminary results indicated that, because of its uncondensed nature, the ALBTH lignin was a good lignin feedstock for hydrogenolysis. Unlike in the LMC reactions, the condensation of the real lignin in biomass under ALBTH conditions was greatly diminished, possibly due to lignin remaining in the solid state, limiting its mobility and accessibility of the benzyl carbocation to the electron-rich aromatic sites for condensation. Reactions using lignin model compounds (LMCs, guaiacylglycerol-β-guaiacyl ether and various aromatic more » monomers) confirmed the formation of the uncondensed moieties and revealed the synergy between LiBr and acid in inducing the crucial intermediate benzyl carbocations, which then led to cleavage of the β- O-4-aryl ether bonds to produce Hibbert's ketones, demethylation to produce benzodioxanes, and condensation reactions. The benzodioxane structure was identified and confirmed for the first time in an acid-depolymerized lignin.
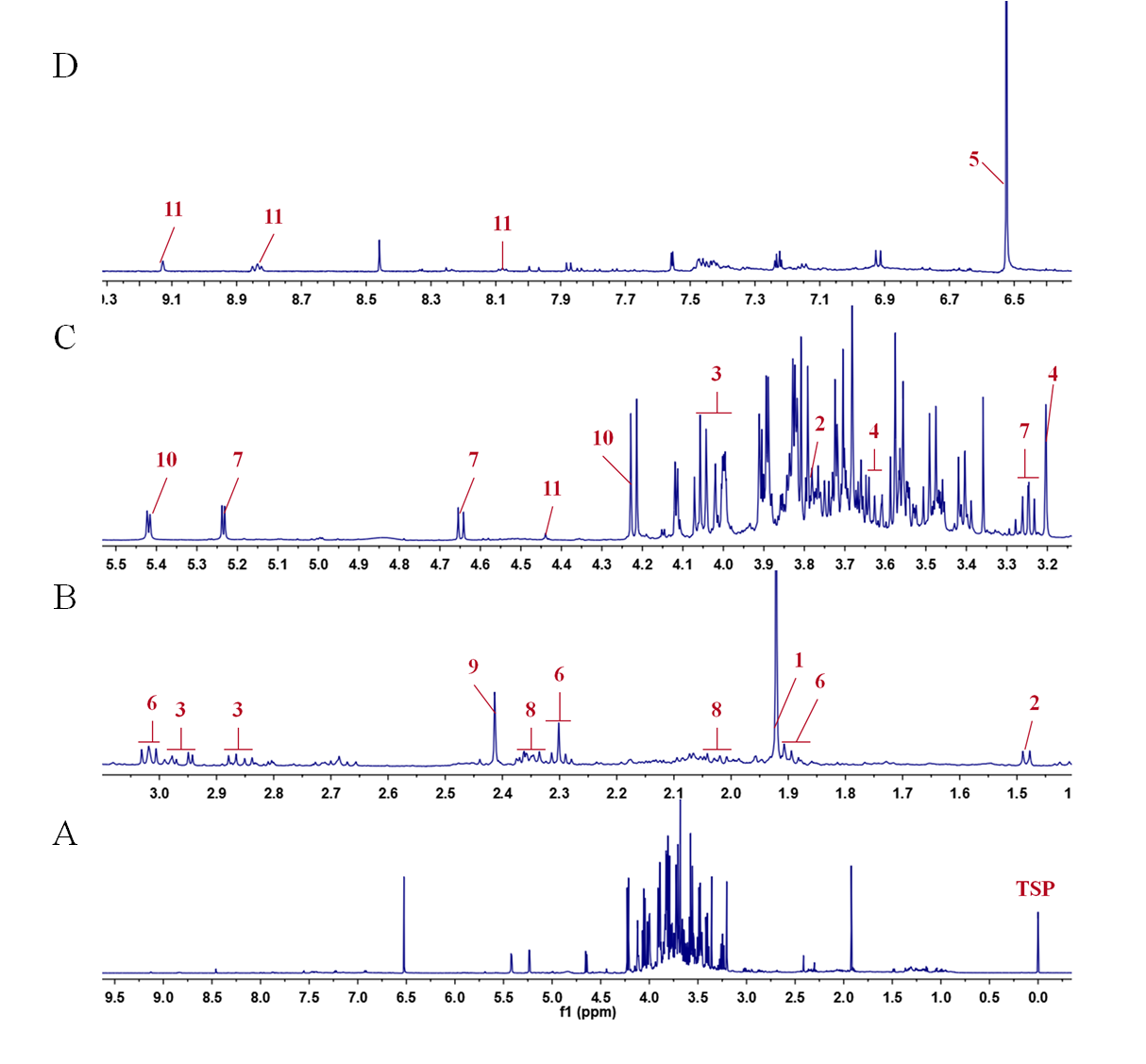
The ALBTH lignins isolated from biomass sources representing the three plant classes (hardwoods, softwoods, and grasses) contained abundant uncondensed moieties (i.e., Hibbert's ketones and benzodioxanes). Here, this study demonstrated that lignin could be efficiently depolymerized in the solid state with minimal condensation and separated from biomass with high purity by dissolving and hydrolyzing cellulose and hemicelluloses, using an acidic lithium bromide trihydrate (ALBTH) system under mild conditions (with 40 mM HCl at 110 ☌). Publication Date: Research Org.: Great Lakes Bioenergy Research Center (GLBRC), Madison, WI (United States) Sponsoring Org.: USDOE Office of Science (SC), Biological and Environmental Research (BER) OSTI Identifier: 1543278 Grant/Contract Number: SC0018409 FC02-07ER64494 AC05-000R22725 Resource Type: Accepted Manuscript Journal Name: Science Advances Additional Journal Information: Journal Volume: 4 Journal Issue: 9 Journal ID: ISSN 2375-2548 Publisher: AAAS Country of Publication: United States Language: English Subject: 59 BASIC BIOLOGICAL SCIENCES 37 INORGANIC, ORGANIC, PHYSICAL, AND ANALYTICAL = ,

